 Interestingly, the molecules of most natural fatty acids have an even number of carbon atoms. A mnemonic phrase for the C 10 to C 20 natural fatty acids capric, lauric, myristic, palmitic, stearic and arachidic is: " Curly, Larry & Moe Perform Silly Antics" (note that the names of the three stooges are in alphabetical order). As shown in the following table, these long-chain carboxylic acids are usually referred to by their common names, which in most cases reflect their sources. The fatty acids are important components of the biomolecules known as lipids, especially fats and oils. Simple alkyl carboxylic acids, composed of four to ten carbon atoms, are liquids or low melting solids having very unpleasant odors. For this group of compounds one such phrase is: " Oh My Such Good Apple Pie".Ĭarboxylic acids are widespread in nature, often combined with other functional groups. Common names, such as these can be troublesome to remember, so mnemonic aids, which take the form of a catchy phrase, have been devised. Simple dicarboxylic acids having the general formula HO 2C–(CH 2) n–CO 2H(where n = 0 to 5) are known by the common names: Oxalic (n=0), Malonic (n=1), Succinic (n=2), Glutaric (n=3), Adipic (n=4) and Pimelic (n=5) Acids. Some examples of both nomenclatures are provided below. In other cases, common names make use of the Greek letter notation for carbon atoms near the carboxyl group. 
Some common names, the amino acid threonine for example, do not have any systematic origin and must simply be memorized. If you are uncertain about the IUPAC rules for nomenclature you should review them now. Substituted carboxylic acids are named either by the IUPAC system or by common names. These two nomenclatures are illustrated in the following table, along with their melting and boiling points. The characteristic IUPAC suffix for a carboxyl group is " oic acid", and care must be taken not to confuse this systematic nomenclature with the similar common system. In the IUPAC system of nomenclature the carboxyl carbon is designated #1, and other substituents are located and named accordingly. In this case, the change in chemical and physical properties resulting from the interaction of the hydroxyl and carbonyl group are so profound that the combination is customarily treated as a distinct and different functional group.Īs with aldehydes, the carboxyl group must be located at the end of a carbon chain. 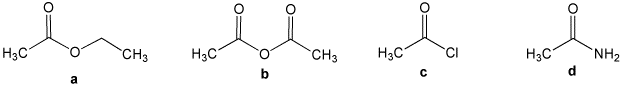
Other combinations of functional groups were described previously, and significant changes in chemical behavior as a result of group interactions were described (e.g. It is often written in condensed form as –CO 2H or –COOH. As may be seen in the formula on the right, the carboxyl group is made up of a hydroxyl group bonded to a carbonyl group. The carboxyl functional group that characterizes the carboxylic acids is unusual in that it is composed of two functional groups described earlier in this text. Carboxylic Acid Reactivity Carboxylic Acids 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
